April 2025

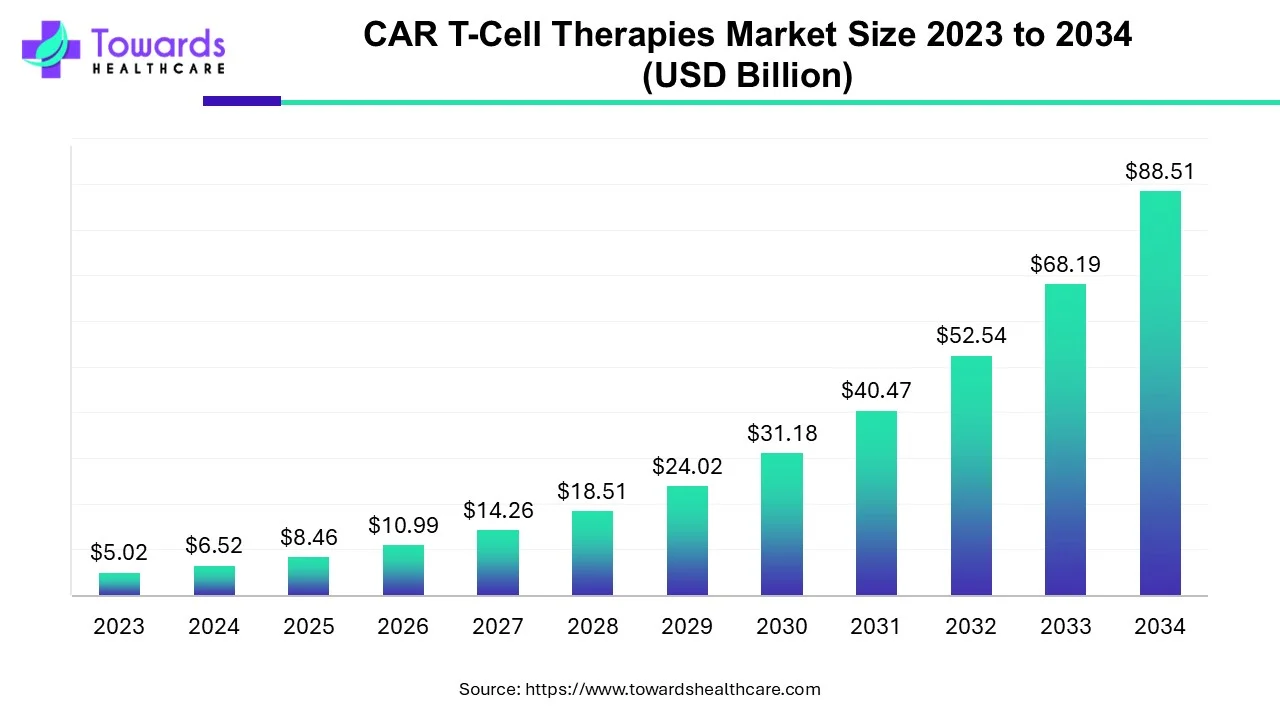
The global CAR T-Cell therapies market is estimated to grow from USD 6.52 billion in 2024 at 29.8% CAGR (2024-2034) to reach an estimated USD 88.51 billion by 2034, as a result of extensive clinical trials, the rising prevalence of cancer, and favorable government policies.
The United States and China reported most of the trials on Clinicaltrials.gov while Europe lags behind in terms of CAR T-Cell clinical trials.
CAR T-cell therapies have significant transformative potential in the field of cancer treatment as they harness the power of the immune system to fight cancer. These therapies involve genetically engineering T cells to express a chimeric antigen receptor that specifically targets a tumor antigen, providing a highly personalized approach to cancer treatment.
CAR-T therapy is a type of cell therapy in which immune T cells are modified genetically to attack the cancer cells by means of injecting chimeric antigen receptors. It is also referred to as a "miracle anticancer drug" due to its high response rate. It does, however, have a complicated manufacturing process that includes collecting the patient's T cells at a medical facility and then culturing them in a good manufacturing practice (GMP) facility. The advantages of CAR T-cell therapies over standard medicines include the use of patients’ immune systems to destroy cancer cells, early recovery, and shorter treatment times. Furthermore, CAR T-cell therapy has a long survival time in the body because it has the ability to recognize and target cancer cells even if cancer recurs. CAR T-cell therapy is primarily used to treat lymphoma, acute lymphocytic leukemia, and multiple myeloma. Due to their numerous advantages over traditional drugs, CAR T-cell therapy products have a high adoption rate, resulting in market growth.
Artificial intelligence (AI) has immense potential to revolutionize the applications of CAR T-cell therapies. Integrating AI into CAR T-cells could assist researchers in developing novel therapies. AI and the Internet of Things (IoT) can enable more precise tailoring of CAR T-cell therapy depending on the patient’s needs. This streamlines the therapeutic process, elevating treatment efficacy and mitigating adverse effects. AI can also be used to automate the production process of CAR T-cells, enhancing efficiency, accuracy, and reproducibility. AI can identify appropriate patients for therapy, help monitor therapy progression, and predict treatment responses. The patients can be monitored through the use of smart devices and wearables to analyze the data collected and respond quickly.

Clinical data has demonstrated the effectiveness of CAR T therapies, particularly in treating relapsed or refractory liquid tumors such as acute lymphoblastic leukemia (ALL) and diffuse large B-cell lymphoma (DLBCL). The approval of CAR T-cell therapies for these indications has marked a significant milestone in cancer treatment and has encouraged further research into the potential of these therapies for other tumor types. CAR T-cell therapy has been shown to induce long-lasting remissions in some patients, even those with advanced or relapsed disease who have failed other treatments. However, it is important to note that CAR T-cell therapy is still a relatively new and complex treatment approach, and there are still challenges and limitations that need to be addressed. The growing industry interest in CAR T-cell therapies is driven by promising clinical data and the potential to develop highly personalized cancer treatments.
Following are some of the CAR-T cell therapy products involved in clinical trials reported as of April 2023
| Drugs | Company | Phase | Indication |
| Descartes-11 | Cartesian Therapeutics | Phase II | Multiple myeloma |
| KTE-X19 | Gilead Sciences | Phase II | Hematological malignancies; Solid tumors |
| MB-CART2019.1 | Miltenyi Biomedicine | Phase III | Non-Hodgkin's lymphoma |
| CT 103A | Innovent Biologics | Pre-registration | Multiple myeloma |
| JWCAR029 | JW Therapeutics | Pre-registration | Diffuse large B cell lymphoma |
| P-BCMA-101 | Poseida Therapeutics | Phase II | Multiple myeloma |
| JCAR017 | Juno Therapeutics | Pre-registration | Non-Hodgkin's lymphoma |
| BPX-603 | Bellicum Pharmaceuticals | Phase I/II | Solid tumours |
| Orvacabtagene autoleucel | Juno Therapeutics | Phase I/II | Multiple myeloma |
CAR T-cell therapy is an innovative approach to cancer treatment that involves genetically engineering a patient's T-cells to recognize and attack cancer cells. The CAR T-cells are designed to target a specific protein found on the surface of cancer cells, allowing them to selectively kill cancer cells while leaving healthy cells intact. CAR T-cell therapy is a highly personalized approach to cancer treatment that requires the extraction of a patient's own T-cells, which are then genetically modified in a laboratory setting to express a chimeric antigen receptor (CAR) that targets the cancer cells. The modified T-cells are then expanded in number and infused back into the patient's bloodstream. Once in the body, the CAR T-cells can recognize and attack cancer cells that express the targeted protein on their surface, leading to the destruction of cancer cells and potentially offering a cure for some patients. During the forecast period, an increase in cancer incidence is anticipated to drive the growth of the global CAR T-Cell treatment market. The main cause of death worldwide is cancer. Almost 400000 kids are diagnosed with cancer in children every year. The most prevalent malignancies differ between nations. Cervical cancer is the most common kind in 23 countries.
The success of CAR T-cell therapy has been most notable in the treatment of hematological malignancies, such as leukemia and lymphoma. Clinical trials have shown that CAR T-cell therapy can produce complete remission in a significant proportion of patients who have exhausted other treatment options. Rising clinical trials and increasing product approvals for treating cancers are significant factors augmenting the growth of the CAR T-cell therapy market.
Combining CAR T-cell therapy with other immunotherapies is an area of active research and has shown promising results in preclinical and clinical studies. One approach is to combine CAR T-cell therapy with checkpoint inhibitors, which are drugs that block proteins that prevent T-cells from attacking cancer cells. By blocking these proteins, checkpoint inhibitors can enhance the effectiveness of CAR T-cells. Another approach is to combine CAR T-cell therapy with other types of immune cells, such as natural killer cells or T-cell receptor (TCR) engineered T-cells. These approaches aim to enhance the overall immune response against cancer cells and increase the durability of the response. Additionally, researchers are exploring the use of CAR T-cells as a delivery vehicle for other immunotherapies, such as cytokines or antibodies. This approach aims to enhance the potency and specificity of these therapies by delivering them directly to the tumor site.
The goal of combining CAR T cells with other immunotherapies is to improve the response rate and duration of response, particularly in patients with solid tumors who have shown limited response to CAR T-cell therapy alone. In addition, studies have shown promising results in combining CAR T cells with checkpoint inhibitors like pembrolizumab or atezolizumab. These agents can help to enhance the activity of the CAR T cells by blocking the immune system's ability to suppress their function. The combination has shown improved response rates and longer progression-free survival in patients with certain types of solid tumors, including lung cancer and breast cancer.
Other combinations that have been studied include CAR T cells with other types of immune cells, such as natural killer (NK) cells or cytokine-induced killer (CIK) cells, and targeted therapies like tyrosine kinase inhibitors. These combinations have shown potential in preclinical studies and early-phase clinical trials, but more research is needed to determine their safety and efficacy in larger patient populations.
By drug type, the axicabtagene ciloleucel segment held a dominant presence in the CAR T-cell therapies market in 2024. Axicabtagene Ciloleucel is used as a second-line agent for the treatment of large B-cell lymphoma that has failed conventional treatment. The rising incidences of B-cell lymphoma govern the segment’s growth.
By drug type, the tisagenlecleucel segment is predicted to witness significant growth in the market over the forecast period. Tisagenlecleucel is used to treat adults with certain types of B-cell non-Hodgkin lymphoma and people up to 25 years old with B-cell acute lymphoblastic leukemia. The demand for tisagenlecleucel increases due to its safety profile, especially in young children and infants, and greater survival rates for patients.
By indication, the lymphoma segment led the global CAR T-cell therapies market in 2024. Lymphoma is a type of cancer that affects the lymphatic system. The increasing incidences of lymphoma and growing research and development activities potentiate the segment’s growth. The lack of potential small molecules for the treatment of lymphoma necessitates the development of CAR T-cell therapies.
By indication, the acute lymphocytic leukemia segment is projected to expand rapidly in the market in the coming years. Acute lymphocytic leukemia (ALL) is a type of cancer affecting blood and bone marrow. The growing incidence and prevalence of acute lymphocytic leukemia and its severity promote the segment’s growth.
By end-user, the hospitals segment held the largest share of the CAR T-cell therapies market in 2024. The segmental growth is attributed to the availability of suitable infrastructure, favorable reimbursement policies, and suitable capital investments. A growing number of hospitals are now adopting affordable CAR T-cell therapies, especially in developed and developing countries.
By end-user, the cancer treatment centers segment is anticipated to grow with the highest CAGR in the market during the studied years. These centers have specific instruments, infrastructure, and expertise to provide CAR T-cell therapies. The increasing number of cancer treatment centers owing to the rising incidences of cancer also propel the segment’s growth.
North America dominated the global CAR T-cell therapies market in 2024. The state-of-the-art research and development facilities, the presence of key players, and favorable reimbursement policies drive the market. The rising incidences of cancer due to the increasing geriatric population also govern market growth. According to the American Cancer Society, around 6,550 new cases of ALL were estimated in the U.S. in 2024, with about 1,330 deaths. The market is also driven by advanced healthcare infrastructure in North America. It is reported that around 311 hospitals in the U.S. are accredited by the Foundation for the Accreditation of Cellular Therapy (FACT) to provide CAR T-cell therapy. Similarly, there are four hospitals in Ontario itself, Canada, that offer CAR T-cell therapy.
Asia-Pacific is projected to host the fastest-growing market in the coming years. The increasing incidences of cancer, increasing investments and collaborations, and growing research and development activities drive the market. Favorable government policies of Asia-Pacific countries favor market growth. The initiatives by the Indian Government, such as Atmanirbhar Bharat and Made in India, support the indigenous development of CAR T-cell therapies. The increasing number of clinical trials also contributes to the market growth. Recently, China surpassed the U.S. in the number of clinical trials, indicating a rapid expansion and growing leadership in CAR T-cell therapy.
For instance,
Regenerative medicines such as cell therapies, gene therapies, and tissue engineering play a vital role in healing tissues and organs and restoring function lost due to aging, disease, damage, or defects. The demand for regenerative medicine is increasing due to the inability of small molecules to effectively treat the disease due to its complexity. The growing research and development activities related to regenerative medicine, increasing investments, and public-private partnerships contribute to the growth of CAR T-cell therapies in the market. In CAR, T-cell therapy introduces a gene to a person’s T cells. CAR T-cell therapies are widely used in hematological malignancies such as lymphoma and leukemia. The favorable regulatory framework supports the launch of novel regenerative medicines, boosting market growth.
Despite these limitations, CAR T-cell therapy is still an exciting and promising approach to treating cancer. There are ongoing efforts to address these limitations and improve the effectiveness and safety of CAR T-cell therapy. For example, researchers are exploring the use of combination therapies that combine CAR T-cell therapy with other treatments to enhance its effectiveness and durability.
The limitations of CAR T-cell therapy underscore the need for continued research and development in this area. The high cost of CAR T-cell therapy, for instance, highlights the importance of developing new manufacturing and production processes that can lower the cost of these treatments. The limited scope of CAR T-cell therapy also underscores the need for ongoing research into new targets and approaches that can be used to treat a wider range of cancers.
The side effects and safety concerns associated with CAR T-cell therapy highlight the importance of careful patient selection and monitoring, as well as ongoing efforts to improve the safety and efficacy of these treatments. This includes the development of new CAR designs that can improve the specificity of CAR T-cell therapy and reduce the risk of off-target effects.
The limited durability of CAR T-cell therapy highlights the need for ongoing research into the underlying biology of cancer and the mechanisms of resistance to CAR T-cell therapy. This includes the development of new combination therapies that can enhance the effectiveness and durability of CAR T-cell therapy, as well as the identification of new targets and approaches that can be used to overcome resistance to these treatments.
Thus, while CAR T-cell therapy has limitations, it is a promising approach to treating cancer that has already shown significant clinical benefits in certain patient populations. The ongoing research and development in this area will be critical in addressing the limitations of CAR T-cell therapy and realizing its full potential as a cancer treatment.
The rapid pace of innovation in the field of CAR T-cell therapy, and recent product approvals are positive signs for the future of cancer treatment. Extensive R&D has played a significant role in overcoming the limitations of CAR T-cell therapy and improving its efficacy. Strategic collaborations among market players are also fostering the growth of the CAR T-Cell therapy market. The development of novel CAR constructs, new methods for T-cell expansion, and the discovery of new tumor-specific antigens have all contributed to the advancement of CAR T-cell therapy. In addition, improvements in manufacturing processes, such as the use of automated closed systems and cryopreservation, have increased the consistency and quality of CAR T-cell products.
Another critical factor in the development of CAR T-cell therapy is the increase in product approvals. As more products gain approval, the market for CAR T-cell therapy is growing rapidly, creating a competitive landscape that is driving innovation and further research. The FDA has already approved six CAR T-cell therapies for the treatment of certain types of blood cancers, and numerous other CAR T-cell therapies are in clinical trials.
Furthermore, researchers are exploring the use of CAR T-cell therapy in solid tumors. Unlike blood cancers, solid tumors have a more complex microenvironment that can limit the effectiveness of CAR T-cell therapy. Overcoming these challenges will require further research, but the potential benefits of CAR T-cell therapy in solid tumors are substantial.
| Generic Name | Brand Name | Target Antigen | Targeted Disease |
| Tisagenlecleucel | Kymriah | CD19 | B-cell acute lymphoblastic leukemia (ALL) |
| Axicabtagene ciloleucel | Yescarta | CD19 | B-cell non-Hodgkin lymphoma (NHL) |
| Brexucabtagene | Tecartus | CD19 | Mantle cell lymphoma (MCL) |
| Lisocabtagene maraleucel | Breyanzi | CD19 | B-cell non-Hodgkin lymphoma (NHL) |
| Idecabtagene vicleucel | Abecma | BCMA | Multiple myeloma |
| Ciltacabtagene autoleucel | Carvykti | BCMA | Multiple myeloma |
Paolo Siciliano, Associate Partner and Life Sciences Expert at PA Consulting envisioned that the field of CAR T-cell therapies will see considerable advancements over the upcoming decade. He predicted certain advancements including CAR-T for solid tumors, extended applications into non-oncology indications, and technological advancements. He also added that CAR T-cell can show positive results in the allogeneic space in future.

By Drug Type
By Indication
By End User
By Geography
April 2025
April 2025
April 2025
April 2025