April 2025

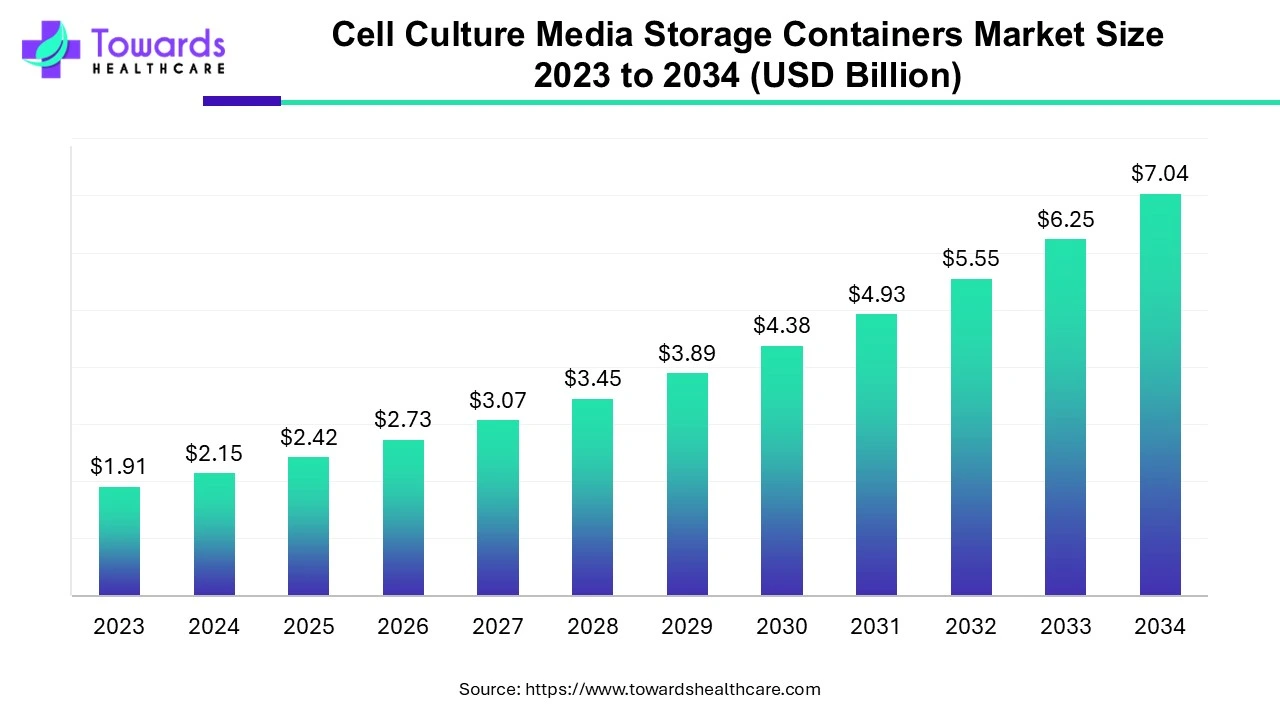
The global cell culture media storage containers market size is calculated at US$ 2.15 in 2024, grow to US$ 2.42 billion in 2025, and is projected to reach around US$ 7.04 billion by 2034. The market is expanding at a CAGR of 12.59% between 2024 and 2034. Cell culture is highly essential and various pharmaceutical and biotechnological processes due to which the demand for cell culture media storage containers is high in these industries.
Specialized containers made to store and maintain the sterility and quality of cell culture medium are known as cell culture media storage containers. These containers, which include bottles, flasks, and bags, are made from premium, non-reactive materials like polypropylene or polyethylene and are available in a variety of sizes and styles. The media stays stable and uncontaminated since they are usually pre-sterilized and have tight-fitting lids that stop leaks and contamination. Reliable cell culture experiments and laboratory research depend on these containers.
When assessing possible end-product containers for cellular treatments, a number of factors are considered. In addition to physically holding the product, a suitable final product container should also offer sufficient stability and physical protection for the product across its whole existence. This affects leachable and extractable materials, temperature stability, light protection, particle levels, and basic container integrity maintenance during production, storage, and transportation. For the supplied container to be successful commercially, it must function in the production process and be sufficiently scalable.
Integration of AI is not very significant in the production of cell culture media storage containers. However, AI can be used for monitoring the overall process of supply chain management, production, and distribution of the containers. Apart from that, AI can be used for monitoring the media and cell culture conditions in the bottles and bags. This can prevent manual checking, improve efficiency, and chances of human errors.
Growing Investments in Research
Technological improvements in the materials and design of cell culture medium storage containers are the result of research investments. The need for the containers is further increased by these developments, which include the creation of specific coatings or materials that improve cell adherence and growth. These developments also make the containers more dependable and effective for research applications. Furthermore, a wider variety of studies and experiments—the bulk of which include cell cultures—are produced by growing funding in life science research. These cultures must be kept and maintained under exacting conditions, which drives the need for premium, specialized cell culture medium storage containers, which are an essential part of the infrastructure for contemporary life sciences research.
Risk of Contamination
Despite the fact that these storage containers have many advantages, the market for storage containers is severely hampered by the possibility of contamination during storage. Contaminants like bacteria, fungus, or viruses can enter the culture media and cause contamination if storage containers are not well sealed or if they are not thoroughly cleaned before use.
Emphasis on Personalized Medicine
The cell culture media storage containers market is expected to develop in the future due to the growing emphasis on customized medicine. Precision medicine, another name for personalized medicine, is a method of providing healthcare that tailors procedures and treatments to each patient's unique needs. The need for more accurate and effective therapies, together with developments in genomics and precision healthcare technology, are the main causes of this increased emphasis on customized medicine. Storage containers for cell culture medium are essential to personalized medicine because they preserve and sustain cell cultures, which makes it possible to create customized treatments and therapies for individual patients.
By product, the storage bags segment held the largest share of the cell culture media storage containers market in 2023. Finding a suitable solution for practically any volume is feasible because of the wide range of off-the-shelf final product bags that are frequently customizable. Because of this, bagged finished goods may be especially desirable for production processes with low numbers but high volumes. When it comes to beginning materials for a large-scale manufacturing process that is already employing "single-use" ingredients and is otherwise a closed system, bags can be incredibly helpful. The additional area for labeling that comes with packaged items is often a nice feature.
By product, the storage bottles segment is estimated to grow at a significant rate in the cell culture media storage containers market during the forecast period. Better ergonomic characteristics, simplicity of use, and a lower risk of contamination are the goals of the design. The media bottle's small size makes handling and storing it simpler. Typically, media bottles have screw-on tops and are made of glass or plastic that is both chemically and biologically inert. These bottles are ideal for long-term storage of volatile organic materials or non-hazardous compounds.
By application, the biopharmaceutical production segment dominated the cell culture media storage containers market in 2023. Biopharmaceuticals currently account for almost 25% of newly authorized medications. One area of the pharmaceutical industry that is expanding quickly is biopharmaceuticals. Recombinant vaccines, hormones, growth factors, cytokines, blood factors, therapeutic enzymes, and monoclonal antibodies (mAbs) are some of the major categories of biopharmaceuticals. Among these, mAb sales have grown at an astounding average rate of 35% over the last three years. Our primary therapeutic areas—cardiovascular, renal, and metabolic, as well as respiratory and immunological, and vaccine and immune—are handled by biopharmaceuticals.
By application, the tissue engineering & regenerative medicine segment is expected to achieve the fastest CAGR in the cell culture media storage containers market during the predicted timeframe. Examining novel approaches to enhance the medical treatment of the elderly and ill population is a global challenge. Among several approaches to this objective, tissue engineering and regenerative medicine (TERM) has steadily developed into a viable method to satisfy patients' demands in the future. The goal of tissue engineering and regenerative medicine research is to replace or regenerate damaged or diseased tissues, organs, and cells. This is a difficult undertaking, but one that offers enormous potential for the practice of medicine. Technologies being studied include stem cell treatments, immunological therapy, and biomaterial/cell constructions for mending different tissues and organs.
By end-use, the pharmaceutical & biotechnology companies segment registered its dominance in the cell culture media storage containers market in 2023. These companies are expanding their research and development and introducing new products for the betterment of healthcare. During the research, development of therapeutics, testing of drugs, and development of biopharmaceuticals, pharmaceutical and biotechnology companies develop cell lines and use various cell culture media storage containers in various shapes and sizes.
By end-use, the CROs & CMOs segment is predicted to witness the fastest growth rate in the cell culture media storage containers market during 2024-2034. Biopharmaceutical businesses have recently relied more and more on contract services that are outsourced for different aspects of their value chain. This is happening in a setting where these businesses are under pressure to reduce expenses and shorten time-to-market in order to address the pandemic's difficulties and cope with financial and resource limitations—all the while having to adhere to laws. Company names include contract manufacturing organizations (CMOs), contract manufacturing and development organizations (CDMOs), and contract research organizations (CROs). Pharma and life science are able to deal with and adjust to the sharp rise in demand, the quickly shifting demands of international drug manufacturing partners, and the growing rivalry from startups vying for market share thanks to these outsourcing partners.
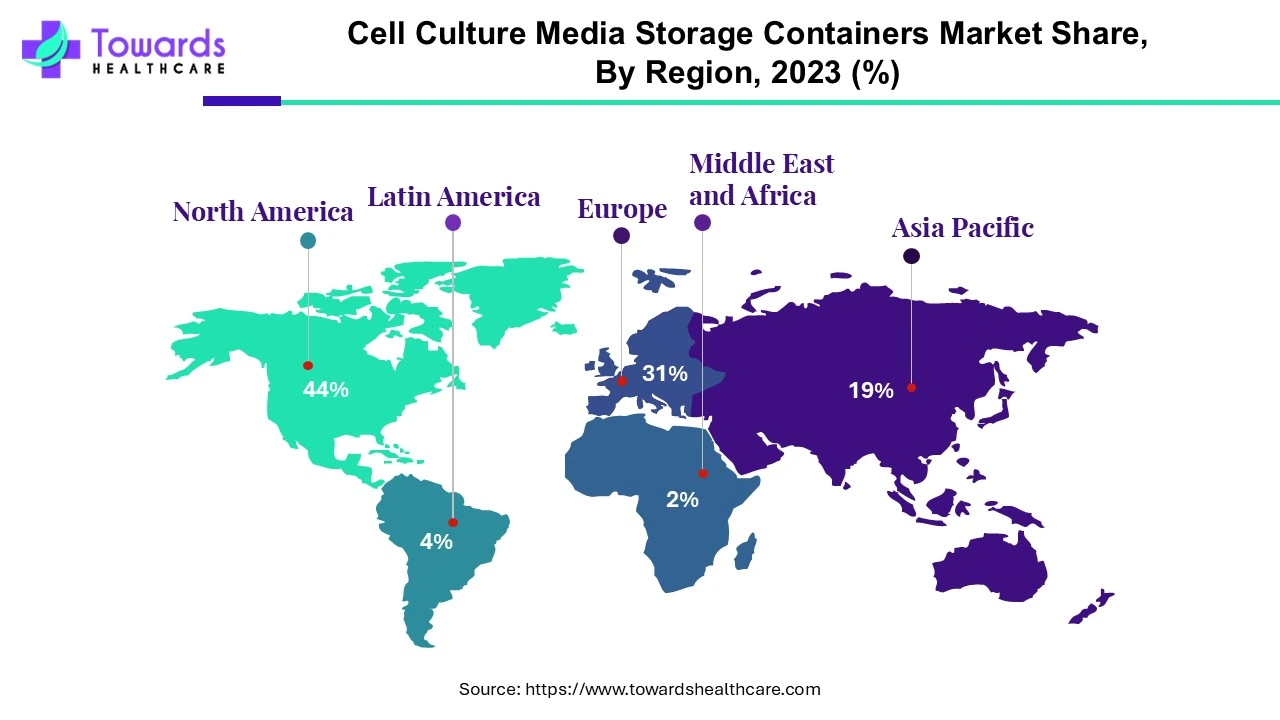
North America dominated the cell culture media storage containers market by 44% in 2023. The growing use of cell culture techniques, which have several benefits including high productivity and low cost, is largely responsible for the increase. The region's relatively higher healthcare expenditure levels, the existence of modern healthcare facilities, the rising need for biologics, and the increasing awareness of storage containers are the factors driving market growth. These elements are expected to drive the growth of the cell culture medium storage container market in North America over the forecast period.
Asia Pacific is estimated to host the fastest-growing cell culture media storage containers market during the forecast period. A number of beneficial policies put in place by the government and industry participants, as well as the opening of new markets, economic growth, and improved healthcare infrastructure, have all contributed to the biopharmaceutical and biotechnology sector's significant expansion. The growing median age of the population and the increased manufacturing of medications, vaccines, and other items are also driving the expansion of the global economy overall. The market for cell culture medium storage containers in this area is expected to expand as a result of these considerations.

In September 2024, being the first manufacturer of cell culture medium to be audited and certified in accordance with the new EXCiPACT GMP Guideline is an honor for us. According to Ivan Donzelot, Head of Integrated Supply Chain Operations for Merck's Life Science business, this standard is essential to ensuring the safety, effectiveness, and quality of cell culture medium in the usage of therapeutic components and is evidence of the value Merck provides to its life science clients.
By Product
By Application
By End-use
By Region
April 2025
April 2025
April 2025
April 2025