April 2025

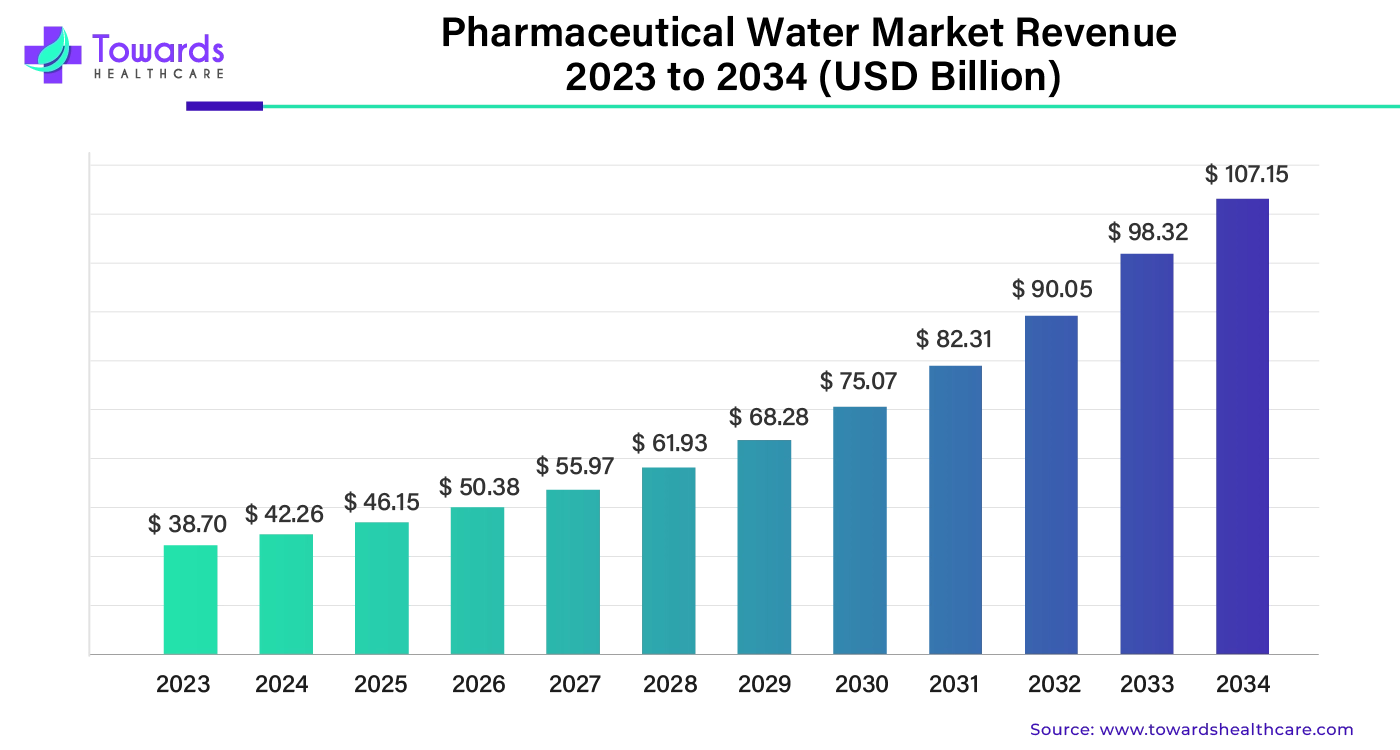
The global pharmaceutical water market size was estimated at US$ 38.70 billion in 2023 and is projected to grow to US$ 107.15 billion by 2034, rising at a compound annual growth rate (CAGR) of 9.25% from 2024 to 2034.
Water is a very important component used in the pharmaceutical industry for a wide range of purposes. Pharmaceutical water is developed for different purposes, which is why the water comes in different grades. These grades include general, drinking water, bulk purified water, bulk highly purified water, bulk water for injections, and other grades of water. For medical purposes, pharmaceutical water is used as a raw material and solvent in the formulation, processing, and manufacturing of various pharmaceutical products, APIs (active pharmaceutical ingredients), analytical reagents, and intermediates. Pharmaceutical water is also used for reconstitution of products, as an excipient, during synthesis, and for the production of final products. For non-medical activities, pharmaceutical water is used for cleaning & rinsing vessels, primary packaging materials, and equipment.
Artificial intelligence has a significant influence on water quality; it is essential for observing, evaluating, and preserving the integrity of our water supplies. Artificial intelligence (AI) technologies transform water treatment systems by anticipating variations in demand and modifying treatment procedures correspondingly. The smart control systems improve overall water treatment standards, lower energy usage, and increase efficiency. Intelligent filtration systems powered by AI adjust to changing water quality circumstances. With the help of sensors and AI algorithms, these systems optimize filtering procedures according to the particular pollutants that are present, guaranteeing complete purification and lowering the possibility of dangerous materials. By anticipating equipment failures and planning maintenance tasks, artificial intelligence (AI) enables autonomous maintenance of water purification systems.
The pharmaceutical water market is growing due to the growing demand for biosimilars and biopharmaceuticals. Biopharmaceuticals are recombinant proteins, monoclonal antibodies, cell & gene therapies, and vaccines. There is a high demand for these products in the healthcare industry due to rising cancer cases, chronic disease cases, genetic disorders, and rare disorders. These biosimilars and biopharmaceuticals are not only effective but can be customized based on the personalized needs of a patient. Furthermore, these products are even useful in researching and developing new drugs and treatment processes and studying different diseases/conditions. Healthcare professionals can also use these products for patient monitoring and treatment. With so many applications, pharmaceutical companies have started to produce these products on a large scale, which has increased the usage of pharmaceutical water.
Pharmaceutical water should be of very high quality because it is used for the production of various therapeutics and can impact the quality of care. Therefore, pharmaceutical water goes through various testing steps, such as total organic carbon (TOC) test, endotoxin test, bioburden test, WFI test, etc. Conducting all these tests is mandatory to ensure all the safety standards are followed. This costs the organization a lot of resources and financial support. As these tests are unavoidable, companies invest a lot in the testing processes, which restricts the growth of the pharmaceutical water market to a certain level. Organizations can use advanced technologies, artificial intelligence, machine learning, and other technologies to analyze tests better. It can help reduce costs and increase efficiency and accuracy.
The pharmaceutical water market needs better water purification technologies to save resources and sustain the production of pharmaceutical water. Some technologies being used for purification include reverse osmosis, UV oxidation, distillation, electrode ionization, and ultrafiltration. With the growing shortage of water, more efficient systems are required for water purification. The news technologies for TOC monitoring, data analytics for predictive maintenance, remote system access, and automation are some of the technological advancements that can create growth opportunities for the market.
For instance,
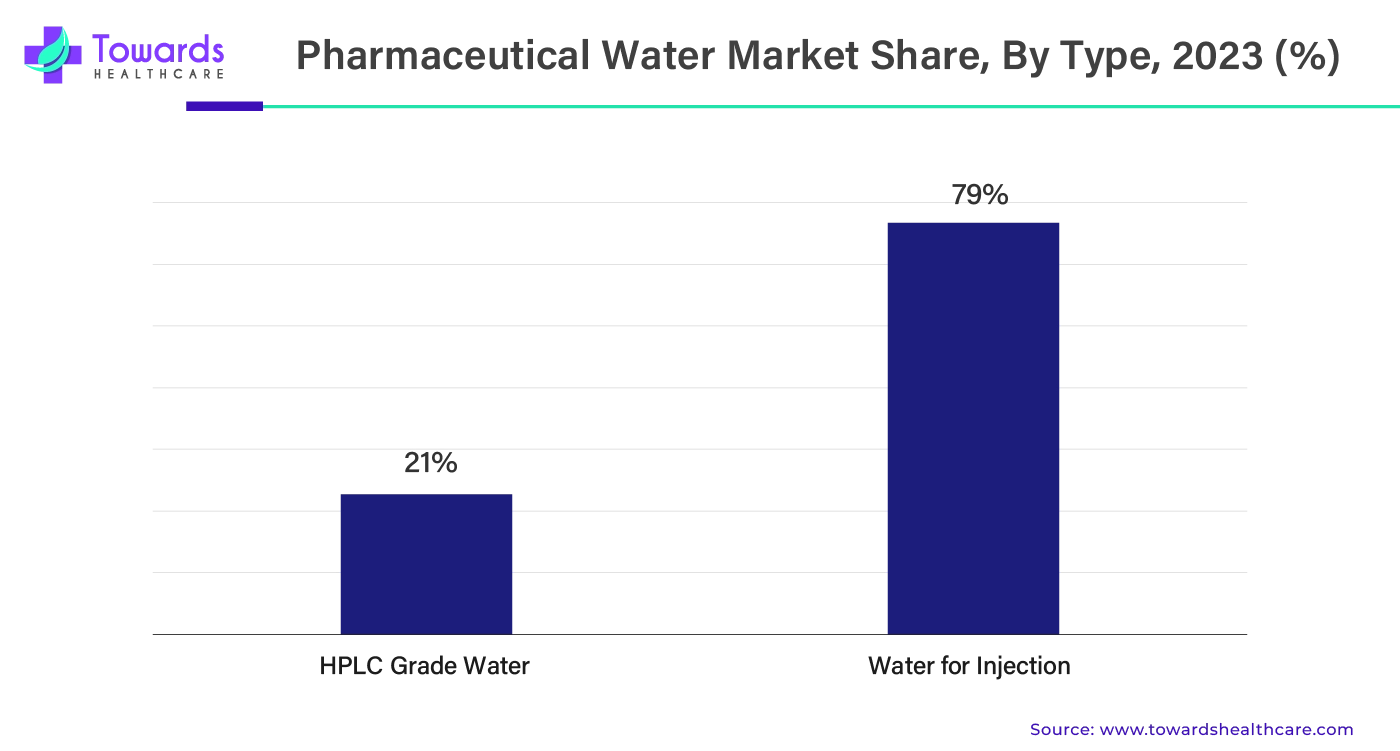
By type, the water for injection segment held the largest share by 79% of the pharmaceutical water market in 2023. Water for injection (WFI) is a high-quality sterile water used to make solutions that are going to be given using injection. WFI is mainly used for medicines that should be protected from damage. WFI is also used as a solvent for better absorption of medicines. There are various standards set by the U.S. Pharmacopeia for the presence of bacteria, organic carbon, and endotoxins.
For instance,
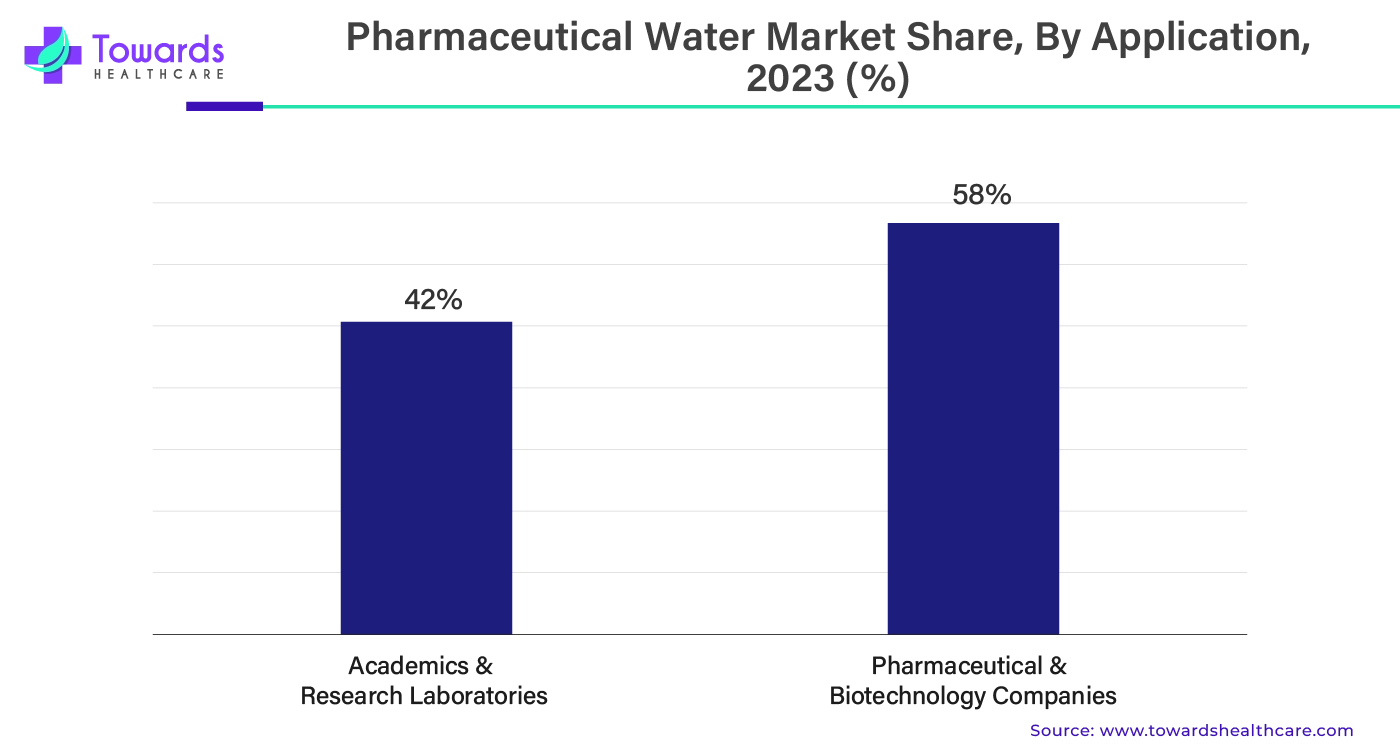
By application, the pharmaceutical & biotechnology companies segment held the majority of the pharmaceutical water market shares by 58% in 2023. There is a continuous demand for research and development in pharmaceutical and biotechnology companies. These demands are increasing due to the increasing demand for better therapeutics, especially for chronic conditions, which are on the rise for various reasons. Apart from this, there are various rare and neglected diseases that do not have any specific cure or medication. Pharmaceutical and biotechnology companies are heavily investing in finding drugs and therapeutics for such diseases. The companies also collaborate with each other and government entities to conduct research and clinical trials. During all these processes, companies utilize a lot of pharmaceutical water. In the future, with the development of new drugs and therapeutics, the large-scale production of these products will increase the demand for pharmaceutical water.
For instance,
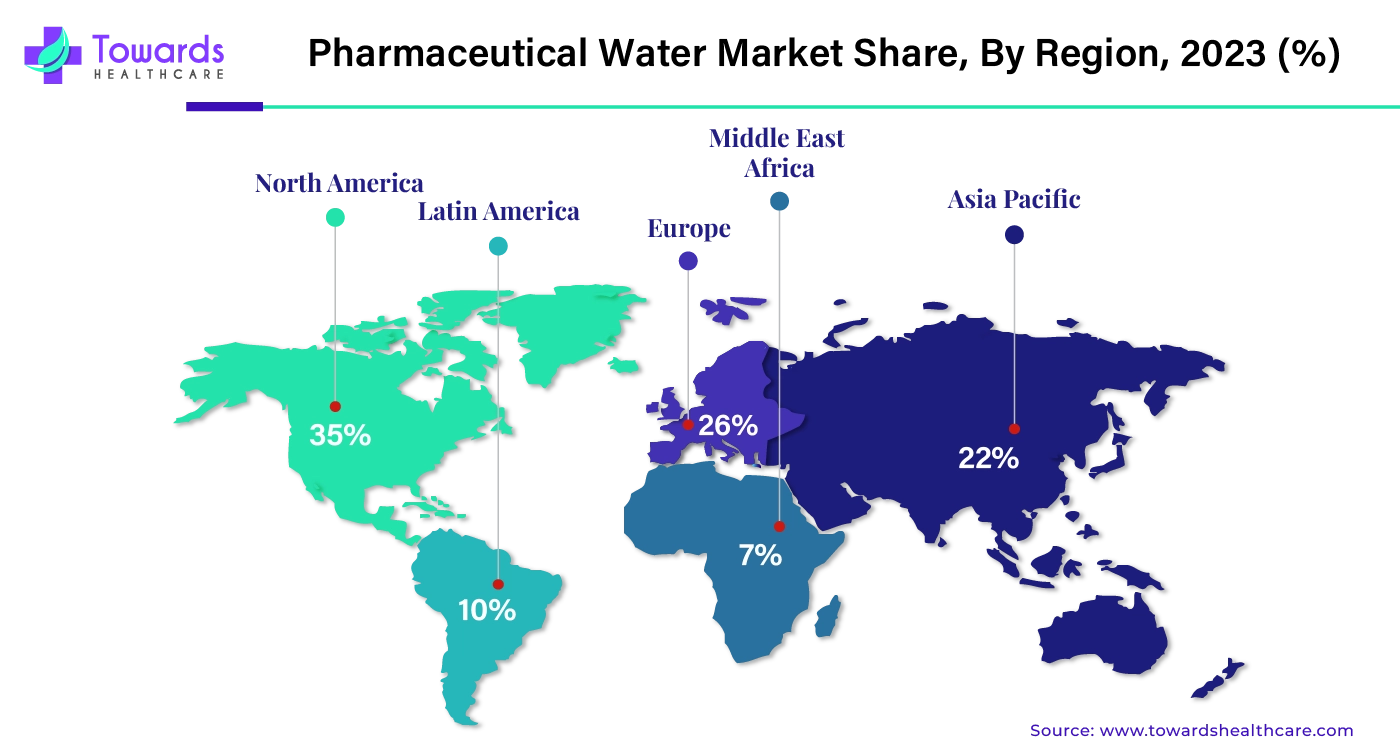
By region, North America dominated the pharmaceutical water market share by 35% in 2023. North America dominates the pharmaceutical, biotechnology, and healthcare industries, which majorly use pharmaceutical water for various purposes. North America is also known for technological advancements which contribute to the growth of the market. The two major countries that promote North America’s growth are the U.S. and Canada. Both countries have a strong presence in the pharmaceutical and biotechnology industries.
The U.S. dominated North America’s pharmaceutical water market in 2023. In 2023, the FDA approved 55 new drugs, out of which 17 were biologics, 9 were TIDES, and 29 were small molecules. According to the International Trade Administration, various countries have invested in the U.S. pharmaceutical and biotechnology industries. These countries are Germany (US$ 6 Billion), Japan (US$ 7 Billion), U.K. (US$ 5 Billion), and Switzerland (US$ 2 Billion). Apart from the U.S., Canada also contributes significantly to the market’s growth.
For instance,
By region, Asia Pacific is expected to grow at the fastest rate of 10.34% during the forecast period. Asia Pacific is strongly growing in the pharmaceutical industry. The major countries contributing to the growth of the pharmaceutical water market are India, China, Japan, and South Korea. India is a major contributor due to growing government and other entities' investment in the pharmaceutical industry.
A solid scientific and technological foundation, expanding government backing, a sizable local market, and reasonably priced production have all contributed to India's pharmaceutical industry's rise to prominence as a global leader in research and innovation. Over the fiscal year 2021–2022, Pharmaceuticals generated $42.34 billion in revenue. Generics, over-the-counter, bulk vaccines, contract research & manufacturing, biosimilars, biologics, and bulk medications are some of the major sectors of the Indian pharmaceutical industry.
The pharmaceutical business stands out in that it uses around twenty-three percent of the water utilized globally. India projects that the need for healthcare services will rise due to an aging population, an increase in chronic illnesses, and a fourfold growth in healthcare spending, reaching $200 billion by 2030.
India's pharmaceutical industry is ranked third in the world in terms of volume and is projected to be worth $50 billion. This country sends pharmaceuticals and medications to more than 200 countries worldwide, accounting for 3.5% of total exports.
PLI for Pharmaceuticals: Created with a budget of INR 15,000 Cr until FY 2027–2028, the initiative aims to boost investment and output in the pharmaceutical sector while also encouraging product diversification toward high-value goods.
The purpose of the Production Linked Incentive (PLI) Scheme is to promote domestic manufacturing of critical key starting materials (KSMs), drug intermediates, and active pharmaceutical ingredients (APIs) across the country. Until FY 2029–2030, the plan called for an investment of INR 6,940 Cr across six years, with financial incentives ranging from 5% to 20% over the base year.
India’s Vision Pharma 2047 Goals
| Company Name | Asahi Kasei |
| Headquarters | Tokyo, Japan, Asia Pacific |
| Recent Development | In June 2024, Kasei declared that it has started offering a membrane system for producing WFI, or water for injection, a sterile kind used for injectable preparation. The membrane system was created by utilizing the system design and development capabilities of MicrozaTM hollow-fiber membrane for liquid product filtering and water treatment as an alternative to traditional distillation procedures for manufacturing WFI. This technology makes it possible to produce WFI for less money and with less CO2 emissions by lowering the requirement to create steam. |
| Company Name | Veolia Water Technologies |
| Headquarters | Paris, France, Europe |
| Recent Development | In February 2024, Veolia Water Technologies unveiled the most recent version of PolarisTM, one of its top-performing high-capacity steam generation and water distillation systems, to further serve the pharmaceutical sector. By European, Japanese, and U.S. Pharmacopoeia standards, the skid-mounted PoIarisTM 2.0 Multiple Effect Distiller (MED) and PolarisTM 2.0 Pure Steam Generator (PSG) systems have been designed to consistently deliver the necessary volumes of water for injection (WFI) and pure steam using a variety of standard options. |
By Type
By Application
By Region
April 2025
April 2025
March 2025
March 2025