April 2025

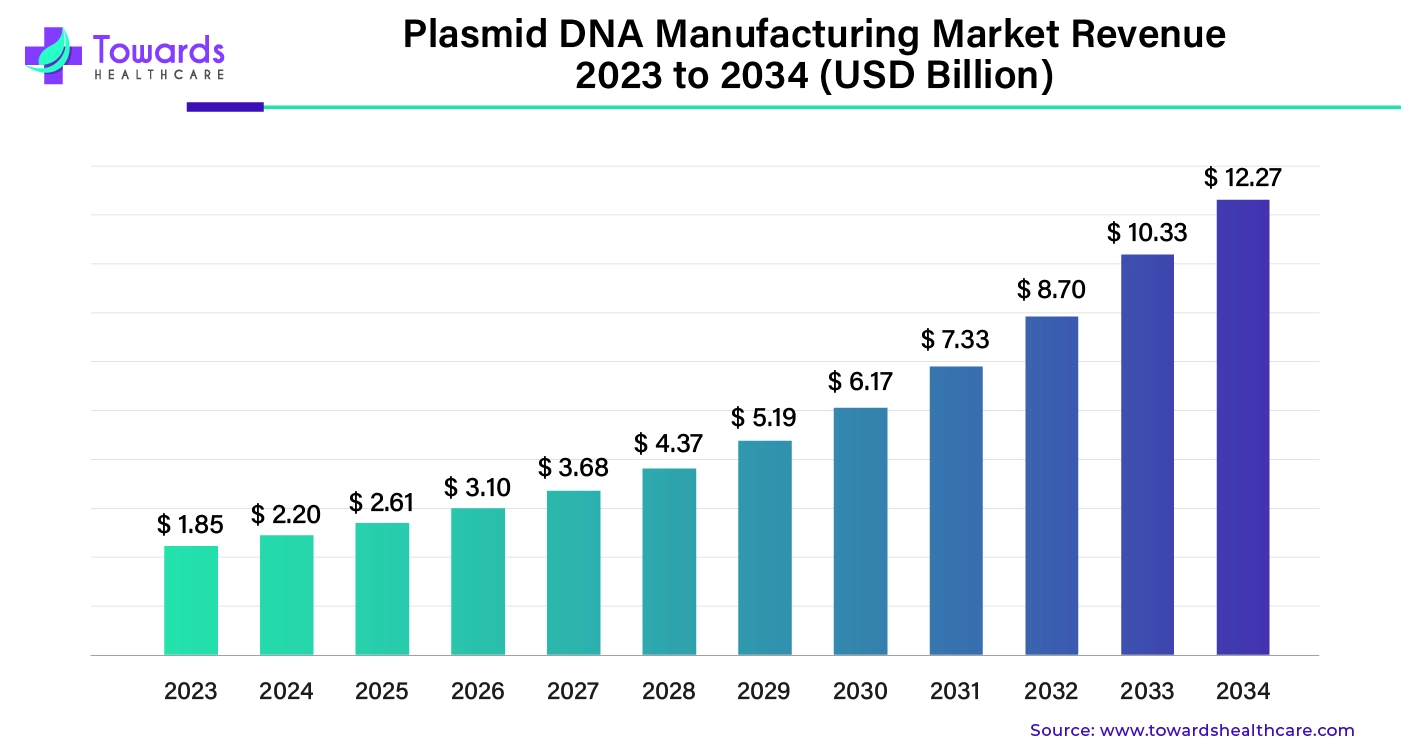
The global plasmid DNA manufacturing market was estimated at US$ 1.85 billion in 2023 and is projected to grow to US$ 12.27 billion by 2034, rising at a compound annual growth rate (CAGR) of 18.77% from 2024 to 2034. Plasmid DNA is highly valuable in conducting research, studying diseases, studying gene mutations, repairing genetic material, and developing GMOs for various healthcare-related purposes. All these applications promote the growth of the market.
The small and circular extra DNA in bacteria and some other microorganisms is called plasmid DNA. During cell division, plasmid DNA makes its copy and passes from one cell to another. This property of plasmid DNA is used by researchers and scientists for genetic modification of viruses, bacteria, and other cells. Plasmid DNA is used in producing vaccines, pharmaceutical proteins, antibodies, hormones, antibiotics, gene therapy, gene transfer, studying genetic disorders, studying the impact of external factors on genetic materials, and so on. DNA plasmid has a lot of applications in healthcare, and with growing demand for pharmaceuticals, antibiotic resistance in pathogens, a growing number of infectious diseases, and cancer, plasmid DNA manufacturing is going to be in high demand in the future.
Plasmid DNAs are in high demand and are produced on a large scale. However, using traditional subjective process control causes highly unstable plasmid yields. To mitigate this challenge, the use of artificial intelligence (AI) and machine learning holds great potential. AI is useful in gene editing as it can achieve affordability, precision, and efficiency. Artificial intelligence is also used in designing. Plasmid DNAs for better stability and success. Patient data can be made easy using AI, and the analyzed data can be used to develop precise and personalized gene therapies, which increases the success rate of treatment.
For instance,
| Company Name | Bionova Scientific |
| Headquarter | U.S., North America |
| Recent Development | In June 2024, Bionova Scientific, the US branch of Asahi Kasei Medical, is a full-service CDMO for biologics. The corporation has decided to establish a new division that will provide services employing plasmid DNA and construct a facility in Texas with the express intent of serving this purpose. The company invested $100 million to expand its plasmid DNA (pDNA) production line. A brand-new, 100,000-square-foot development and production facility is the project's main feature. |
| Company Name | Thermo Fisher Scientific |
| Headquarters | U.S., North America |
| Recent Development | In June 2024, Thermo ScientificTM KingFisherTM PlasmidPro Maxi Processor* (PlasmidPro), the only fully automated maxi-scale plasmid DNA (pDNA) purification technology, was unveiled today by Thermo Fisher Scientific Inc., the global leader in supporting research. PlasmidPro offers full automation for both mini- and maxi-scale purification, facilitating innovation at scale by supplying high-purity plasmids without the need for manual column preparation or intervention. |
| Consignee_Name | Shipper_Name | Product Description | Sum of Quantity | Sum of Weight |
| A AND B INGREDIENTS INC | Cosucra Group | Starches and starch products | 24403 | 517290 |
| ACHIM IMPORTING CO INC | Nanjing Jinming New Decorative Mate | Other organic chemicals | 44587 | 209772 |
| BEST PARTNER SUPPLY CHAIN INC | Yolotech Co Limited | Other organic chemicals | 38863 | 175135 |
| IBIC INTERNATIONAL GROUP INC | Chongqing Tonghui Gas Co Limited Add | Other organic chemicals | 61754 | 176906 |
| MANILDRA MILLING CORP | Shoalhaven Starches Pty Limited | Starches and starch products | 43200 | 2245285 |
| NATURZ ORGANICS LLC | Naturz Organics Dalian Co Limited | Starches and starch products | 24000 | 496000 |
| OKAYA USA | Okaya Co Limited | Other organic chemicals | 47539 | 6297576 |
| PURCHASER COMMERCE ENTERPRISES LLC | Hongkong Inchoii Co Limited | Other organic chemicals | 27424 | 129702 |
| PLAYTEX MANUFACTURING INC | Kdc/One Swallowfield Station | Other organic chemicals | 42044 | 169389 |
| TRAFON GROUP INC | Grupo Lacteo Del Caribe Gruplac | Other organic chemicals | 34464 | 21406 |
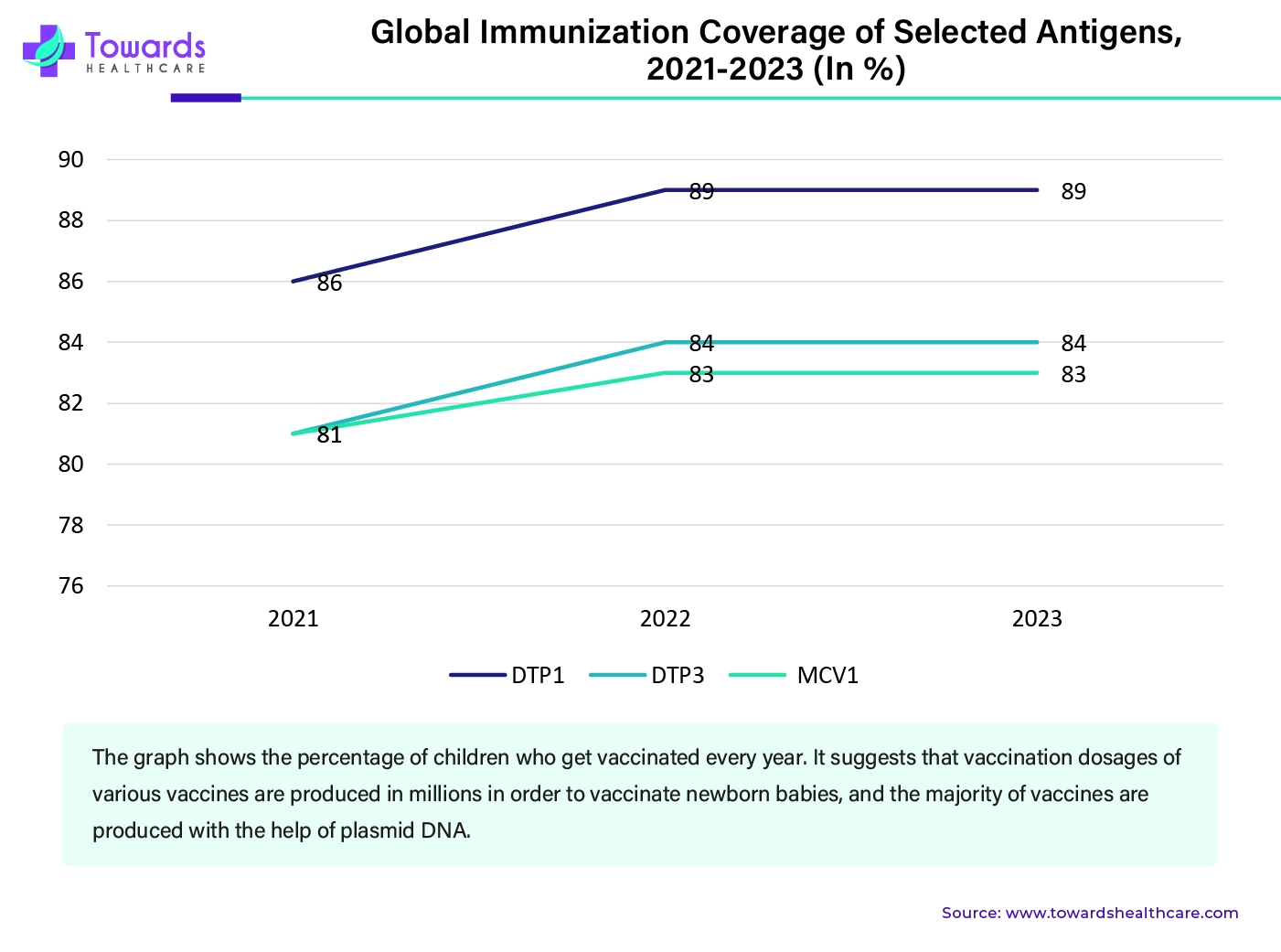
To boost the immune system, vaccines are highly essential. Plasmid DNA is used to make vaccines for various diseases. A lot of vaccines are given at a young age to ensure protection against various diseases. The growing birth rate is one of the major reasons behind the demand for vaccines. According to the United Nations, 385,000 babies are born each day, which is equivalent to 140 million babies every year, which suggests that every year, around 140 million children need different vaccines to boost immunity. Apart from this, a lot of vaccines are given multiple times, which increases the demand. The growing population and need for immunity boost is going to increase the production of plasmid DNA-based vaccines.
Genetic modification raises a lot of ethical concerns because naturally existing genetic material is modified to gain desirable results. Plasmid DNAs are used for inserting and modifying genetic material; therefore, such genetically modified plasmids need to be kept in a highly regulated and controlled environment in order to avoid the risk of losing GMOs in nature. This leakage of GMOs in nature can genetically modify the naturally existing organisms, leading to a huge biothreat. Therefore, governments on national and international levels have very strict regulatory systems to avoid any biothreat. To follow the regulations, extra efforts, time, resources, money, and energy are invested, which becomes a restraining factor for the plasmid DNA manufacturing market.
Cancer is a growing concern and poses a financial burden on the healthcare system. According to the WHO, in 2022, 20 million new cancer cases were anticipated, and by 2050, it is estimated that there will be more than 35 million new cancer cases, which is a 77% increase when compared to 2022. Due to the rise in cancer cases, new plasmid-based therapies are being developed. Plasmid vaccines, gene therapy, and cell therapy are some of the therapies in which plasmids are used. Plasmid DNA also provides the opportunity to develop personalized therapeutics. With the growing use of monoclonal antibodies in cancer treatment, the use of plasmid DNA for making monoclonal antibodies is also increasing.
By product, the viral vector segment held the dominant share of the plasmid DNA manufacturing market in 2023. Viral vectors are used in cell & gene therapies and vaccine administration. Viral vectors are in demand due to their high efficiency in delivering genetic information and excellent targeted delivery of bioactive molecules. Currently, 2-3 billion liters of bioreactor capacity every year is needed for the production of viral vectors, and in the future, the capacity is going to increase even further.
For instance,
By application, the gene therapy segment accounted for the largest share of the plasmid DNA manufacturing market in 2023. Gene therapies are used to fix, replace, and remove genes that cause health issues. Researchers are showing interest in gene therapy due to its potential in treating cancer, rare diseases, and genetic disorders. The number of clinical trials has significantly increased in the last decade. According to the National Institutes of Health, there are more than 5000 gene therapy trials listed with them as of 2023.
For instance,
By disease, the infectious disease segment dominated the plasmid DNA manufacturing market in 2023. Plasmid DNA is used for developing vaccines for infectious diseases. A lot of infectious diseases are highly contagious and can lead to a pandemic like the COVID-19 pandemic. Hence, developing vaccines for infectious diseases is highly needed, and vaccines can be developed using plasmid DNA. Plasmid DNA-based vaccines hold high value in cases where conventional vaccines fail.
For instance,
By region, North America dominated the plasmid DNA manufacturing market share by 44% in 2023. North America leads in research and development associated with healthcare, pharmaceutical, and biotechnology industries. Governments, organizations, healthcare systems, and research institutions contribute to developing new and innovative procedures and products to improve healthcare facilities. With growing cases of cancer, infectious diseases, and genetic disorders, North America has accelerated the research. The government also invests and launches programs to encourage companies to take the initiative. Apart from this, major market players collaborate with each other or government institutions to make collaborative efforts for better results.
The U.S. dominated the plasmid DNA manufacturing market, followed by Canada and North America. The U.S. has advanced technology, better facilities, and resources that help professionals to provide high-quality results. The major players in the U.S. not only supply plasmid DNA but also export the products on a global level, which gives a strong boost to the growth of the market in the U.S. The U.S. also has a strong presence in the healthcare system and the biotech and pharma industry, which uses plasmid DNA for a lot of different purposes.
Apart from this, there is a growing number of cancer and genetic disorders that may need gene therapy. According to research published in 2023 by NIH, it is expected that there will be 1.09 million cancer patients who will receive gene therapy by 2034. It is estimated that the number of patients who are going to be treated with gene therapy in the year 2025 will be 95,696.
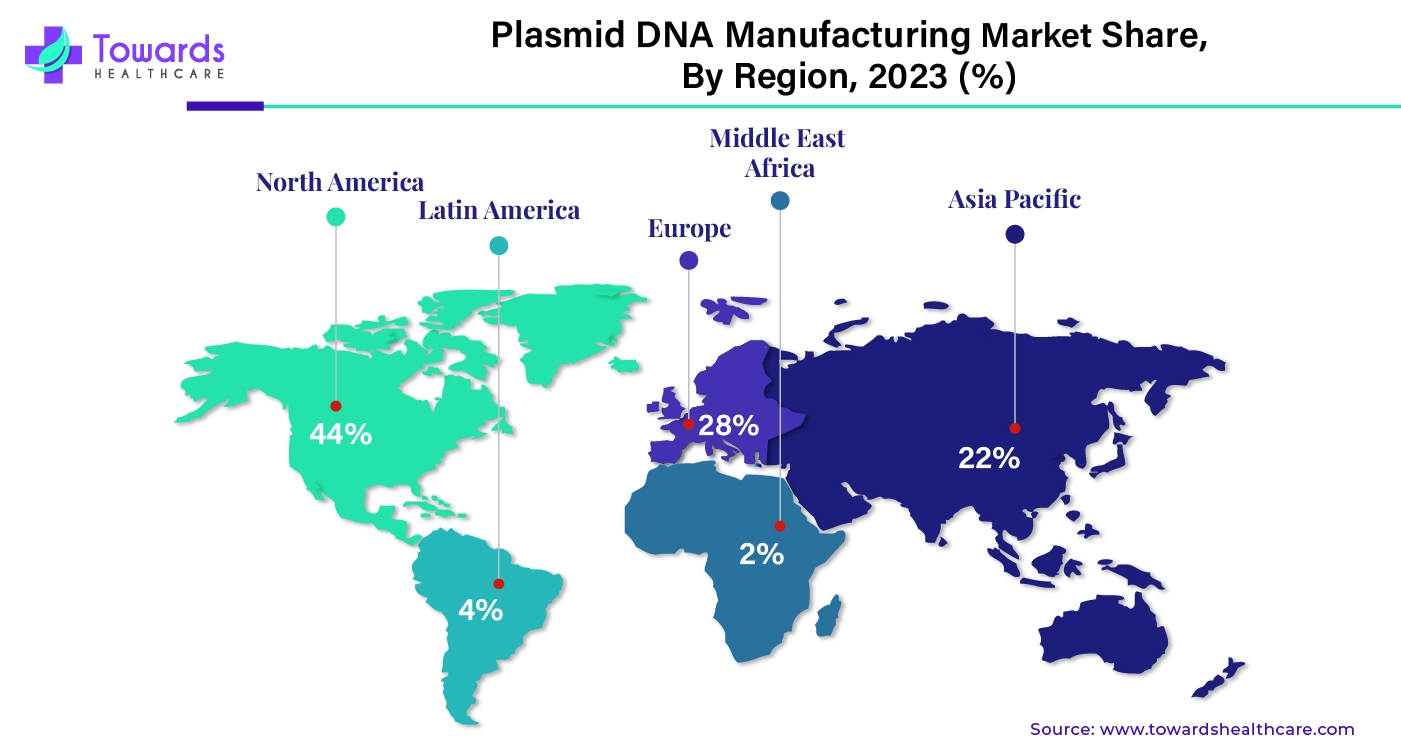
By region, Asia Pacific is expected to grow at the fastest rate during the forecast period. The biotechnology industry is the main user of plasmid DNA. The government and many organizations in Asia Pacific have analyzed the potential of biotechnology in the growth of the region, which is why they are heavily investing in the biotechnology industry. This investment has positively impacted the growth of the plasmid DNA manufacturing market. China, India, and Japan significantly contribute to the growth of the market. The growing population in India and China also promotes that market growth because vaccines and other therapeutics will be needed in large amounts.
In China, urbanization and an aging society are going to demand quality health. It is estimated that there will be 400 million people aged 60 years or above age by 2030. China is highly invested in developing better healthcare solutions, and the government and organizations have taken various initiatives.
The biotechnology industry is also growing strongly in India, which is increasing the use of plasmid DNA for developing vaccines and other therapeutics. When it comes to the size of the Indian biotechnology industry, it holds the 12th position at the global level and the 3rd position in the Asia Pacific. There are more than 800 core biotech companies in India. Some of the factors that have contributed to the growth of the plasmid DNA manufacturing market include the largest production and supply of BCG, DPT, and measles vaccines, the developed world’s first DNA vaccines for COVID-19, and the 2nd highest number of manufacturing plants approved by USFDA.
By Product
By Application
By Disease
By Region
April 2025
April 2025
March 2025
February 2025