April 2025


Principal Consultant

Reviewed By
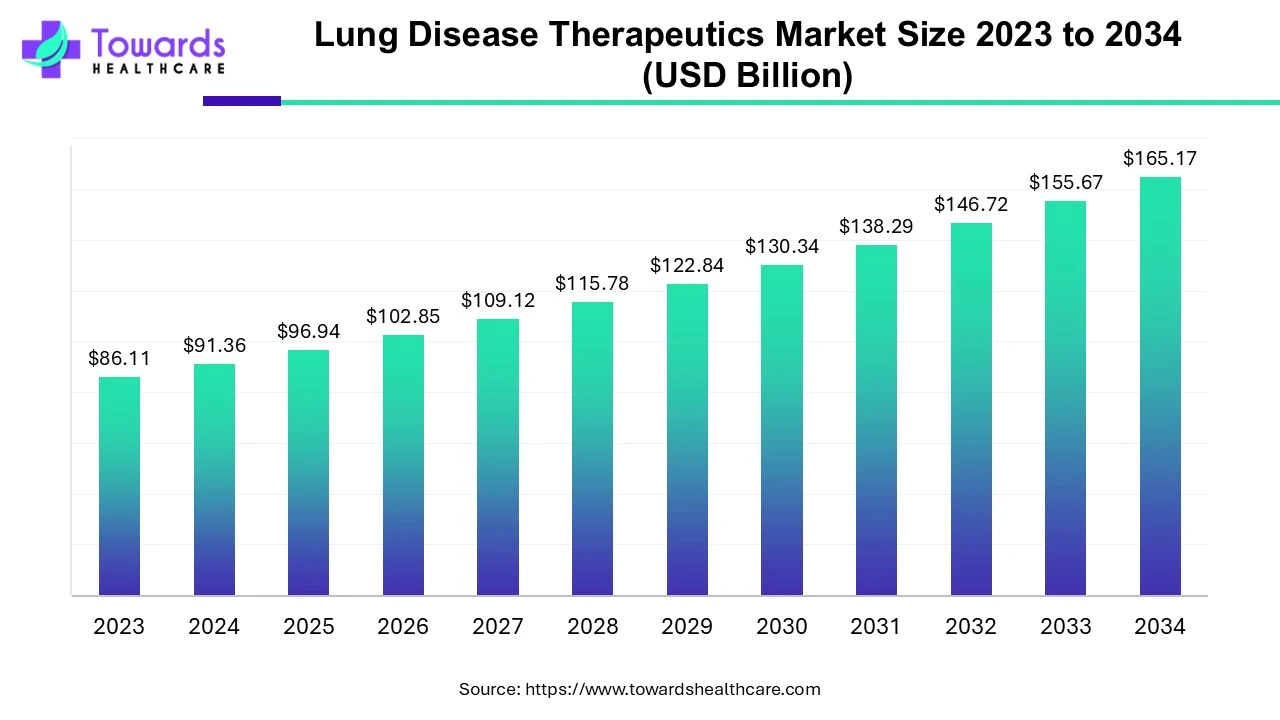
The global lung disease therapeutics market size is projected to reach USD 165.17 billion by 2034, growing from USD 96.94 billion in 2025, at a CAGR of 6.1% during the forecast period from 2025 to 2034, as a result of rising advancement in targeted and personalized therapies and increasing prevalence of associated diseases.
Asthma symptoms are prevalent in approximately 1 in 10 children globally.
The lung disease therapeutics market is witnessing significant growth due to the increasing prevalence of respiratory disorders, including chronic obstructive pulmonary disease (COPD), asthma, cystic fibrosis, and pulmonary arterial hypertension (PAH). These diseases pose a substantial burden on global healthcare systems and require effective therapeutic interventions to manage symptoms, improve patient outcomes, and enhance quality of life.
The market encompasses various therapeutic approaches, including bronchodilators, corticosteroids, targeted therapies, monoclonal antibodies, and gene therapies. These treatments aim to alleviate symptoms, reduce inflammation, restore lung function, and prevent disease progression. The increasing awareness about respiratory health, advancements in diagnostic techniques, and the development of novel treatment options are driving market growth. The rising geriatric population, growing tobacco smoking prevalence, and environmental factors such as air pollution contribute to the high demand for lung disease therapeutics.
Advancements in targeted therapies and personalized medicine are major lung disease therapeutics market drivers. Traditional treatments for lung diseases often focus on symptom management and broad-spectrum therapies. However, the emergence of targeted therapies has revolutionized the treatment landscape, enabling more precise interventions based on individual patient characteristics. The rising prevalence of lung diseases is creating the need for advanced therapeutic interventions thereby augmenting the growth of the lung disease therapeutics market. For instance, according to the Forum of International Respiratory Societies (FIRS)

Targeted therapies involve the use of drugs that specifically target key molecular pathways and cellular mechanisms involved in lung diseases. For example, in the case of non-small cell lung cancer (NSCLC), targeted therapies such as tyrosine kinase inhibitors (TKIs) are designed to block specific genetic mutations or proteins that drive tumor growth. These therapies offer improved treatment outcomes and fewer side effects compared to traditional chemotherapy.
Personalized medicine approaches in lung disease therapeutics utilize genetic testing and biomarker analysis to identify patients who are more likely to respond to specific treatments. For instance, genetic testing can identify patients with EGFR mutations in NSCLC who are more likely to respond to EGFR-targeted therapies. By tailoring treatment strategies based on individual patient characteristics, personalized medicine optimizes therapy selection, improves treatment efficacy, and minimizes adverse effects.
The development of targeted therapies and personalized medicine in lung disease therapeutics is supported by advancements in molecular diagnostics, genomics, and proteomics. These technologies enable the identification of disease-specific biomarkers, genetic variations, and molecular signatures that inform treatment decisions. Furthermore, the integration of artificial intelligence (AI) and machine learning algorithms enhances the analysis of complex molecular data, facilitating the discovery of novel therapeutic targets and personalized treatment regimens.
The increasing focus on inhalation therapies and respiratory drug delivery systems is another major driver in the lung disease therapeutics market. Inhalation therapies offer several advantages over other routes of drug administration, including targeted delivery to the lungs, reduced systemic side effects, and improved patient convenience and compliance. Inhalation therapies involve the use of inhalers, nebulizers, and dry powder inhalers (DPIs) to deliver medications directly to the lungs. These devices enable the efficient delivery of corticosteroids, bronchodilators and other respiratory medications, providing localized treatment and rapid symptom relief.
The development of advanced inhalation technologies and drug delivery systems has expanded treatment options for lung diseases. For instance, the introduction of metered-dose inhalers (MDIs) with spacers has improved medication delivery and reduced the need for hand-lung coordination, benefiting patients with limited dexterity or difficulty in using traditional inhalers.
Additionally, the emergence of smart inhalers, integrated with sensors and connectivity features, has revolutionized respiratory care. Smart inhalers can monitor medication usage, provide reminders, track symptoms, and transmit data to healthcare providers. These devices facilitate better disease management, adherence to treatment plans, and remote patient monitoring, ultimately leading to improved outcomes.
Inhalation therapies and respiratory drug delivery systems are particularly crucial in the management of chronic respiratory conditions like asthma and COPD. These diseases often require long-term maintenance therapy and quick-relief medications. Inhalation therapies offer precise dosing, rapid onset of action, and the ability to adjust dosage based on individual needs, providing optimal disease control and symptom management. Furthermore, the ongoing research and development in inhalation technologies aim to enhance drug delivery efficiency, reduce the frequency of dosing, and improve patient convenience. Novel inhaler designs, formulations, and drug combinations are being explored to overcome the limitations of current inhalation devices and optimize treatment outcomes.
Despite the growth prospects, the lung disease therapeutics market faces significant challenges, with one major restraint being the high cost of therapies and limited accessibility. Many advanced targeted therapies and biologics used in the treatment of lung diseases come with high price tags. The development of these therapies involves extensive research, clinical trials, and regulatory processes, which contribute to their elevated costs. Additionally, the limited patient population for certain rare lung diseases and the need for specialized manufacturing processes further contribute to the high prices of these therapies.
The high cost of lung disease therapeutics poses challenges in terms of affordability, reimbursement, and access to treatment, particularly in low- and middle-income countries. Patients may face financial barriers in accessing these therapies, limiting their ability to benefit from the latest advancements in treatment options.
The pricing and reimbursement policies vary across different healthcare systems and countries, further impacting the accessibility of lung disease therapeutics. The process of obtaining reimbursement coverage for new therapies can be complex and time-consuming, leading to delays in patient access and limited availability in certain regions. Additionally, the lack of healthcare infrastructure and trained healthcare professionals in some regions can hinder the effective delivery of lung disease therapeutics. Limited access to diagnostic facilities, specialized clinics, and healthcare providers with expertise in respiratory care can result in underdiagnosis, inadequate treatment, and suboptimal disease management.
Addressing the high cost and limited accessibility of lung disease therapeutics requires a comprehensive approach involving collaboration between healthcare stakeholders, policymakers, and pharmaceutical companies. Efforts should be made to streamline pricing and reimbursement processes, ensure equitable access to innovative therapies, and promote affordable generic alternatives.
By disease type, the asthma segment held a dominant presence in the market in 2024. The rising prevalence of asthma due to allergies, infections, and air pollution boosts the segment’s growth. Asthma is a major cause of morbidity and mortality, especially among people from low- and middle-income countries. New product launches and growing research and development activities lead to novel asthma therapeutics. Favorable government policies for screening and early detection of asthma also promote the segment’s growth.
By drug class, the corticosteroids segment held the largest share of the lung disease therapeutics market in 2024. Corticosteroids are the most commonly prescribed drugs for lung diseases, as they reduce inflammation and mucus production in the lungs. This helps patients relieve symptoms quickly. They can be provided by both inhalation and injection routes. Inhaled corticosteroids may be used as a regular treatment to help prevent flare-ups and reduce hospitalizations. Hence, they also reduce the risk of exacerbations and improve the quality of life for individuals.
By drug class, the bronchodilators segment is expected to grow at the fastest rate in the market during the forecast period. Bronchodilators are drugs that relax lung muscles and help clear mucus from the lungs. They also help relieve symptoms of asthma, COPD, and other lung conditions. Some common examples of bronchodilators include salbutamol, ipratropium, theophylline, and salmeterol. They are either long-acting or short-acting and are preferred based on the patient’s condition. They are most used in combination with corticosteroids to enhance their therapeutic potential.
By molecule type, the biologics segment held the major share of the lung disease therapeutics market in 2024. Biologics provide targeted effects, thereby reducing systemic side effects. They can easily reach the targeted site by scheduling their release through the development of novel drug delivery systems. They eliminate the risk of potential drug-drug interactions and reduce some harmful effects of small molecules. Thus, biologics reduce the frequency of disease exacerbations and hospital visits. Favorable regulatory frameworks support the development of novel biologics.
By molecule type, the small molecule segment is estimated to show lucrative growth in the market over the studied years. Small molecules can be administered through oral, parenteral, or inhalation routes, increasing medication adherence and patient accessibility. They can favor scheduled releases based on disease requirements. They provide a rapid onset of action and can overcome the risks of biologicals. Biologics can be immunogenic and can induce allergic reactions in sensitized patients, which is not the case for small molecules.
By treatment type, the targeted therapy segment registered its dominance over the global lung disease therapeutics market in 2024. Targeted therapy is majorly used for lung cancer treatment that targets certain abnormalities in tumors. The growing demand for personalized treatment and rising genomics research augment the segment’s growth. These targeted therapies interfere with cancer cells' growth and function. They also reduce the systemic side effects and slow the spread of disease. It is found that up to 30% of lung cancer patients can have molecular targets that respond to targeted therapy.
By treatment type, the immunotherapy segment will gain a significant share of the market over the studied period. Immunotherapy is the most essential treatment regimen for lung diseases to activate the immune system. Severe lung disorders like asthma, COPD, and lung cancer affect the immune response, causing potential adverse effects. Immunotherapy is widely used to treat allergic asthma and COPD. Allergic asthma is the most prominent type, affecting 81% of children and 73% of adults with asthma.
By distribution channel, the hospitals segment led the global market in 2024. The increasing number of hospitalizations due to rising incidences of acute asthma or severe lung disorders propel the segment’s growth. Hospitals have several skilled professionals and favorable reimbursement policies that increase the demand for the treatment of lung diseases in hospitals. They have suitable capital investment to adopt advanced tools and technologies for effective treatment. The presence of suitable infrastructure and the availability of necessary drugs also support the segment’s growth. They are also part of numerous clinical trials, providing efficient treatment before product market approval.
By distribution channel, the online segment is projected to show notable growth in the market during the forecast period. Online services allow patients to purchase their medications, inhalers, or related products in the comfort of their homes. The rising adoption of smartphones and the burgeoning e-commerce sector potentiate the segment’s growth. Customers can buy all types of products based on their preferences from a wide range of options and pricing. Online pharmacies offer free home delivery and online consultancy services, attracting more customers.
North America, specifically the United States, holds a significant share in the lung disease therapeutics market. The region's dominance can be attributed to factors such as a high prevalence of lung diseases, well-established healthcare infrastructure, robust research and development activities, and favorable reimbursement policies. The United States has a considerable burden of respiratory diseases, including COPD, asthma, and lung cancer. The availability of advanced diagnostic tools, a well-developed healthcare system, and a favorable regulatory environment contribute to the growth of the lung disease therapeutics market in the country.

Participants were surveyed regarding whether they had received a diagnosis of chronic obstructive pulmonary disease (COPD), emphysema, or chronic bronchitis from a doctor or other healthcare professional. Furthermore, initiatives focused on smoking cessation, air pollution control, and public awareness campaigns contribute to disease prevention and management.
Europe is also a prominent market for lung disease therapeutics, with countries such as the United Kingdom, Germany, and France leading in terms of market share. The region has a high prevalence of respiratory diseases and significant investments in research and development. Moreover, well-established healthcare systems, strong regulatory frameworks, and a proactive approach to disease management contribute to the growth of the market in Europe.
Asia-Pacific is a rapidly growing market for lung disease therapeutics, fueled by factors such as a large patient population, increasing healthcare expenditure, and a rising prevalence of respiratory diseases. Countries like China, Japan, and India have significant market potential due to the increasing awareness about respiratory health, changing lifestyles, and urbanization contributing to environmental pollution. In terms of the fastest-growing region in the upcoming years, Asia-Pacific is projected to exhibit significant growth. The region's large population, rapid urbanization, increasing healthcare investments, and a growing focus on improving respiratory healthcare infrastructure are expected to drive market expansion.
Latin America and the Middle East & Africa regions are also witnessing growth in the lung disease therapeutics market. The increasing adoption of advanced therapies, improvements in healthcare infrastructure, and initiatives aimed at raising awareness about lung diseases contribute to the market growth in these regions.
The integration of digital health technologies and telemedicine presents a major opportunity in the lung disease therapeutics market. Digital health solutions, including mobile applications, wearables, remote monitoring devices, and telehealth platforms, have the potential to transform the management of lung diseases. Digital health technologies can facilitate remote patient monitoring, real-time data collection, and personalized disease management. Patients can use mobile applications or wearables to track their symptoms, lung function, medication adherence, and lifestyle factors. These data can be shared with healthcare providers, enabling proactive interventions, personalized treatment adjustments, and early detection of disease exacerbations.
Telemedicine platforms allow patients to consult healthcare professionals remotely, reducing the need for in-person visits and enhancing accessibility to specialized care. Telehealth consultations can provide timely advice, education, and support to patients, improving disease understanding, self-management skills, and treatment compliance. The integration of digital health technologies with lung disease therapeutics offers several benefits. It can improve patient engagement, empower individuals to actively participate in their care, and facilitate personalized treatment plans. Moreover, digital solutions can enhance disease surveillance, enable real-world evidence generation, and support research and development efforts.
Market players can capitalize on this opportunity by developing innovative digital health solutions tailored to the specific needs of patients with lung diseases. Mobile applications that provide educational resources, symptom tracking, medication reminders, and inhaler technique guidance can improve self-management and treatment adherence. Furthermore, the integration of data analytics and AI algorithms can enable personalized treatment recommendations, predictive analytics for disease exacerbations, and early intervention strategies. Machine learning algorithms can analyze large datasets, identify patterns, and generate actionable insights to support clinical decision-making and optimize treatment outcomes. Collaborations between pharmaceutical companies, technology developers, and healthcare providers can drive the development and adoption of digital health solutions in the lung disease therapeutics market. Strategic partnerships can facilitate the integration of digital platforms with existing treatment regimens, ensure data security and privacy, and address regulatory considerations.
In conclusion, the lung disease therapeutics market is witnessing a significant growth driven by factors such as the increasing prevalence of respiratory disorders, advancements in targeted therapies and personalized medicine, and the focus on inhalation therapies and respiratory drug delivery systems. However, the high cost of therapies and limited accessibility pose challenges to market growth. North America currently holds a significant market share, with Asia-Pacific projected to be the fastest-growing region in the upcoming years. The integration of digital health technologies and telemedicine presents a major opportunity for market players to enhance disease management, improve patient outcomes, and optimize treatment strategies.

Charlotte Allerton, Head of Discovery and Early Development at Pfizer, commented on their collaboration with Ampersand Biomedicines and Montai Therapeutics to develop therapeutics for obesity and non-small cell lung cancer (NSCLC) that Ampersand’s innovative platform, Pfizer’s deep expertise in cardiometabolic health and cancer drug discovery, and Montai’s computational tools can unlock novel approaches to targets. He added that the company aims to utilize those unique approaches to drive the next wave of breakthroughs for patients.

By Disease Type
By Drug Class
By Molecule Type
By Treatment Type
By Distribution Channel
By Geography
April 2025
March 2025
February 2025
September 2024