February 2026

The global cancer biologics market size is expected to increase from USD 119.41 billion in 2025 to USD 232.02 billion by 2034, growing at a CAGR of 7.66% throughout the forecast period from 2025 to 2034.

The cancer biologics market includes pharmaceuticals made from living things or their parts that are intended to target particular molecules implicated in the development and spread of cancer. Vaccines, cytokines, cell-based immunotherapies, and monoclonal antibodies are examples of these biological therapeutics. In comparison to traditional treatments, they are less toxic and provide tailored mechanisms of action as well as the possibility of customized treatment plans. The market for cancer biologics is expected to grow due to developments in biotechnology and oncology research, which are providing novel treatment options for different forms of cancer and enhancing patient outcomes and quality of life. Clinical trials are the sole way to access several forms of biological therapy. One of the busiest areas of cancer research is biological treatment.
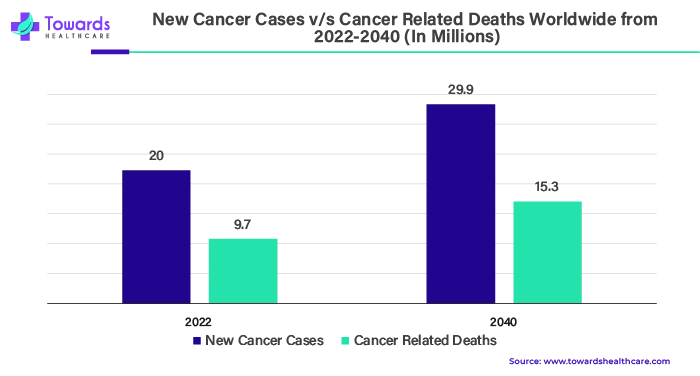
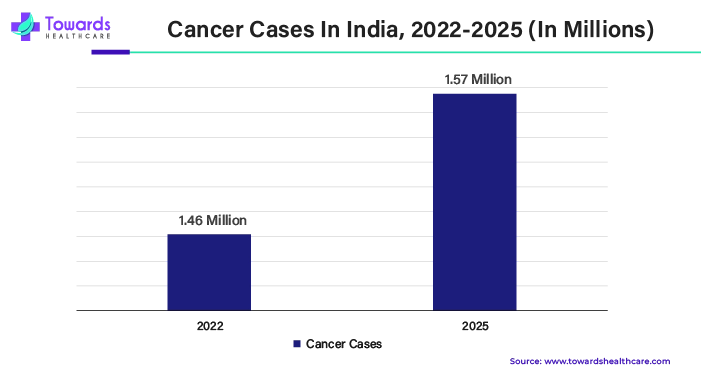
According to WHO predictions, 20 million new cases of cancer and 9.7 million deaths from the disease will occur in 2022. It was estimated that 53.5 million individuals would still be living five years after being diagnosed with cancer. One in five people will experience cancer at some point in their lives. Roughly 1 in 9 men and 1 in 12 women die from the condition. Lung cancer is the most common cancer, accounting for 2.5 million new cases worldwide, or 12.4% of all new cases. Prostate cancer (1.5 million cases, 7.3%), stomach cancer (970,000 cases, 4.9%), colon cancer (1.9 million cases, 9.6%), and female breast cancer (2.3 million cases, 11.6%) were the next in incidence order.
Because biological therapies selectively target malignant cells and minimize their impact on healthy tissue, they have fundamentally altered the field of oncology. The main focus of this publication is on novel medicines that have advanced the care of specific cancers. Combining different biological therapies is becoming more and more important in cancer therapy. Examples of these combinations include immunotherapy with oncolytic viruses or cancer vaccines.
The development, manufacturing, and treatment expenses of cancer biologics are often higher than those of traditional chemotherapeutic drugs. Biological production necessitates specialized infrastructure, stringent quality control standards, and complex manufacturing processes, all of which drive up production costs. Moreover, the individualized character of many biological medications, including monoclonal antibodies that target particular disorders or biochemical pathways, may result in higher treatment costs. Cancer biologics are expensive, which puts obstacles in the way of payers, healthcare providers, and patients. This restricts access to these drugs and may have an impact on the growth of the cancer biologics market.
Systems for delivering drugs are essential to individualized therapy. These systems allow for increased patient compliance, fewer side effects, and better treatment outcomes by customizing drug administration to each patient's unique characteristics. Customizing medical interventions according to an individual's distinct genetic composition, way of life, and medical background is the goal of personalized medicine. In the areas of neoplasia and chronic diseases, biologic drug delivery systems present exciting opportunities for better treatment outcomes. They can fulfill the need for personalized therapies in drug delivery, customize therapeutic and diagnostic procedures (theranostics) for specific patients, and apply precision medicine to the use of biologics.
The monoclonal antibodies segment dominated the cancer biologics market in 2023. Today, monoclonal antibody-based immunotherapy is regarded as an essential part of cancer treatment, in addition to chemotherapy, radiotherapy, and surgery. A wide range of therapeutically significant modes of action are included in monoclonal antibodies. Furthermore, antibodies have the ability to specifically target tumor cells while also encouraging the development of durable immune responses against tumors. The cancer biologics market will be significantly impacted by the new cancer treatment approaches that have been developed as a result of the versatile qualities of antibodies as a therapeutic platform.
For instance,
The cancer growth inhibitors segment is estimated to grow at the fastest rate during the forecast period. A medication known as a "cancer growth blocker" specifically targets and inhibits the growth factors that cause cancer cells to proliferate. Researchers are examining various approaches to accomplish this. Growth blockers for cancer come in a variety of forms. The kinds of compounds that they block can be used to categorize them.
For instance,
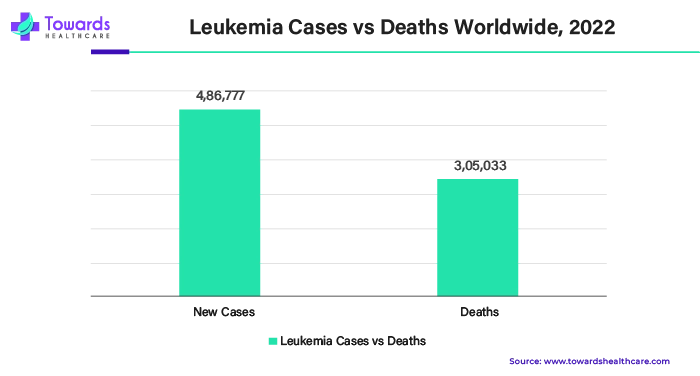
The blood cancer segment held the largest share of the cancer biologics market in 2023. A more recent form of treatment called biologic therapy helps your immune system become more adept at identifying and eliminating cancer cells. It has the ability to strengthen the body's defenses against leukemia. Different types of biologic therapy exist. Chimeric antigen receptor (CAR) T-cell treatment and monoclonal antibody therapy are two biologic medicines used to treat leukemia.
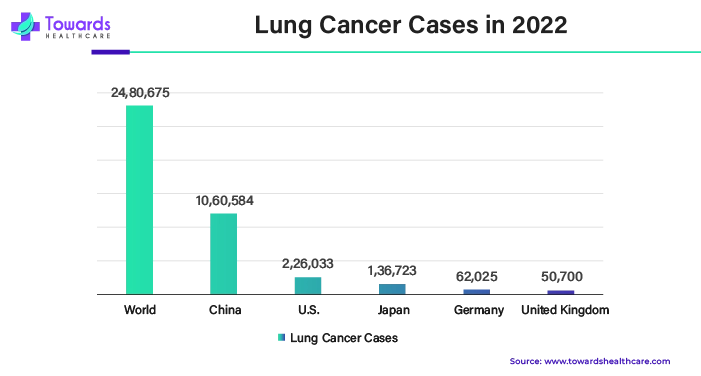
The lung cancer segment is estimated to grow at the fastest CAGR during 2024-2033. One novel and extremely promising approach in conservative oncology is the use of biological therapy in the treatment of lung cancer. Contemporary biologically active preparations possess a high degree of selectivity and adequate antitumor activity, enabling them to target cancer cells specifically. Biological therapy's selective action results in low incidence rates of side effects and treatment acceptability. Biological therapy plays a significant role in the entire treatment of cancer and produces remarkable clinical outcomes.
Furthermore, with an annual incidence of 2.24 million cases and a fatality rate of 1.80 million, LC is the primary cause of cancer-related fatalities globally. The two main histological subtypes of lung cancer are non-small cell lung cancer (NSCLC), which makes up 85% of all lung cancers, and small cell lung carcinoma (SCLC), which accounts for 15% of all lung cancers.
The hospitals segment dominated the cancer biologics market in 2023. When it comes to using biologics and treating cancer, hospitals are the best option. Surgeons, radiologists, oncologists, and other medical specialists with expertise in your particular cancer kind can be found in hospitals. A multidisciplinary approach can drastically reduce problems by bringing together specialists from many professions to work on your treatment plan, which may include cancer surgery.
The cancer centers segment is estimated to grow at the fastest rate during the forecast period. There are huge benefits to having an integrated treatment center. We have specialists under one roof who are accustomed to working together in a multispecialty clinic. Professionals from various fields, such as scientists, physicians, and researchers, work in cancer centers. These individuals study novel, potent cancer therapies. They search for various methods of battling the illness. Healthcare professionals are also taught at a center how to diagnose, prevent, and treat different types of cancer. These centers also conduct clinical investigations and public awareness campaigns. On the other hand, patients receive 24-hour care at a comprehensive cancer center.
For instance,
North America dominated the cancer biologics market in 2023. Well-developed and technologically advanced healthcare infrastructure and well-regulated laws are the main drivers of growth. New biologics are developed more quickly as a result of growing government incentives and expanded research and development efforts in the field of cancer biologics. The quantity of biotechnology and pharmaceutical businesses in this area also encourages the expansion of the regional market.
The U.S. market is growing because of the increasing incidence of cancer and the availability of favorable reimbursement schemes. In addition, groups, including the Esophageal Cancer Awareness Association (ECAA), American Cancer Society, and American Breast Cancer Foundation (ABCF), are initiating initiatives to support patients, family members, survivors, caregivers, and anybody else who may be at risk from the illness. Consequently, growth in the regional cancer biologics market is anticipated during the course of the forecast period.
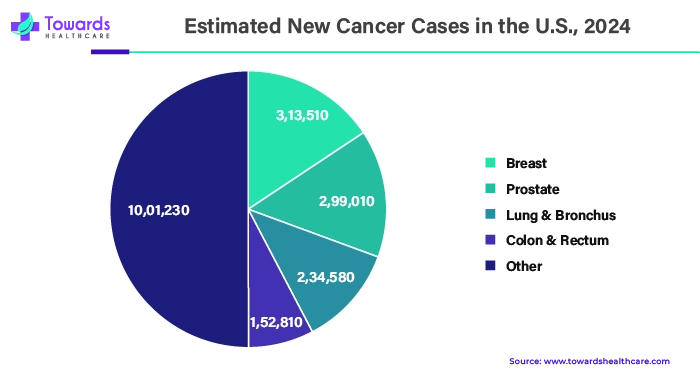
Asia Pacific is expected to grow at the fastest rate during the forecast period. There has been a steady rise in cancer cases in Asia-Pacific, which can be attributed to a number of variables, including aging populations and changing lifestyles. The need for efficient cancer treatments, including biological medications, has increased due to the rise in cancer incidence. The size of the cancer biologics market in Asia-Pacific is also significantly influenced by the region's expanding healthcare infrastructure. The nations in the area have built state-of-the-art hospitals, cancer centers, and research facilities, among other healthcare infrastructure projects, at great financial cost. The development, production, and distribution of biological medications for the treatment of cancer are made possible by this expanded infrastructure, enhancing patient access to these treatments.
Respondents highlighted India's growing domestic demand, bio-investment, and potential for expanded exports to developed countries" are the main drivers of the country's expansion, which is mostly driven by its expanding cancer biologics market. Not only is the global cancer incidence rising, but it is also rising in India. In the past, the majority of cancer cases in India were caused by oral, breast, and cervical malignancies. The country's northeast has a high incidence of cancer, according to trends in the regional distribution.
| Company Name | BioNTech and Duality Biologics |
| Headquarters | BioNTech (Germany, Europe), Duality (China, Asia Pacific) |
| Recent Development | In January 2024, BioNTech partnered with Duality Biologics in China to launch a late-stage trial for their precision medication that targets a particular kind of advanced breast cancer as part of a calculated move to increase its footprint in the oncology space. It was disclosed in a joint announcement by BioNTech and Duality that the late-stage trial will target patients with advanced breast cancer who had low levels of the cell surface receptor HER2. Patients will have their antibody-drug conjugate evaluated in a Phase III trial by BioNTech and Duality. |
| Company Name | Biocon Biologics |
| Headquarters | Karnataka, India, Asia Pacific |
| Recent Development | In June 2024, the European Medicines Agency (EMA) granted the Bengaluru-based biopharma company Biocon Biologics (BBL) permission to manufacture biosimilar Bevacizumab at its recently constructed multi-product monoclonal antibodies (mAbs) medicinal substance facility. Bevacizumab is used to treat a variety of cancer types, such as cervical cancer, non-small cell lung cancer (NSCLC), glioblastoma (a kind of cancerous brain tumor), and colon and rectal cancer (cancer that starts in the large intestine). |
By Drug Class
By Applications
By End-use
By Region
February 2026
February 2026
February 2026
January 2026
